Suprello™ Surgical Wound Matrix
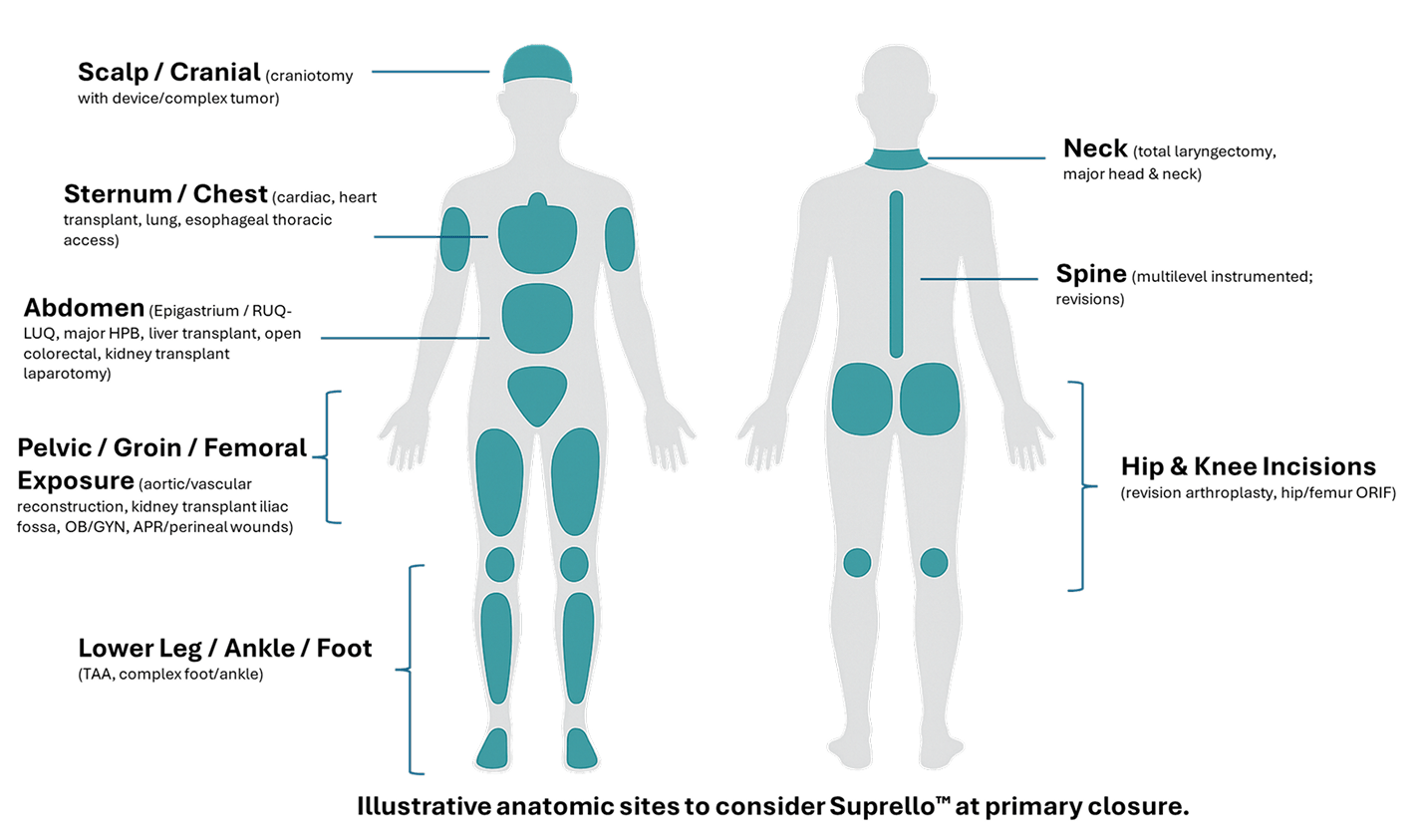
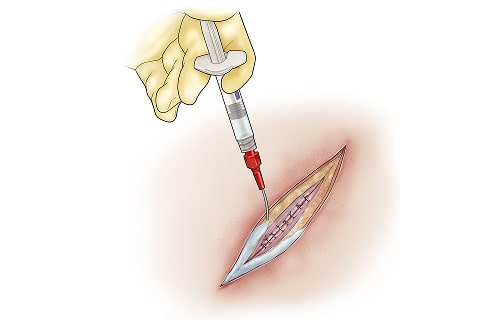
Suprello™ is a peptide-based surgical wound matrix specifically formulated for surgical wounds in high-risk cases. It is designed to support closure and help mitigate the risk of wound dehiscence and associated surgical wound complications.
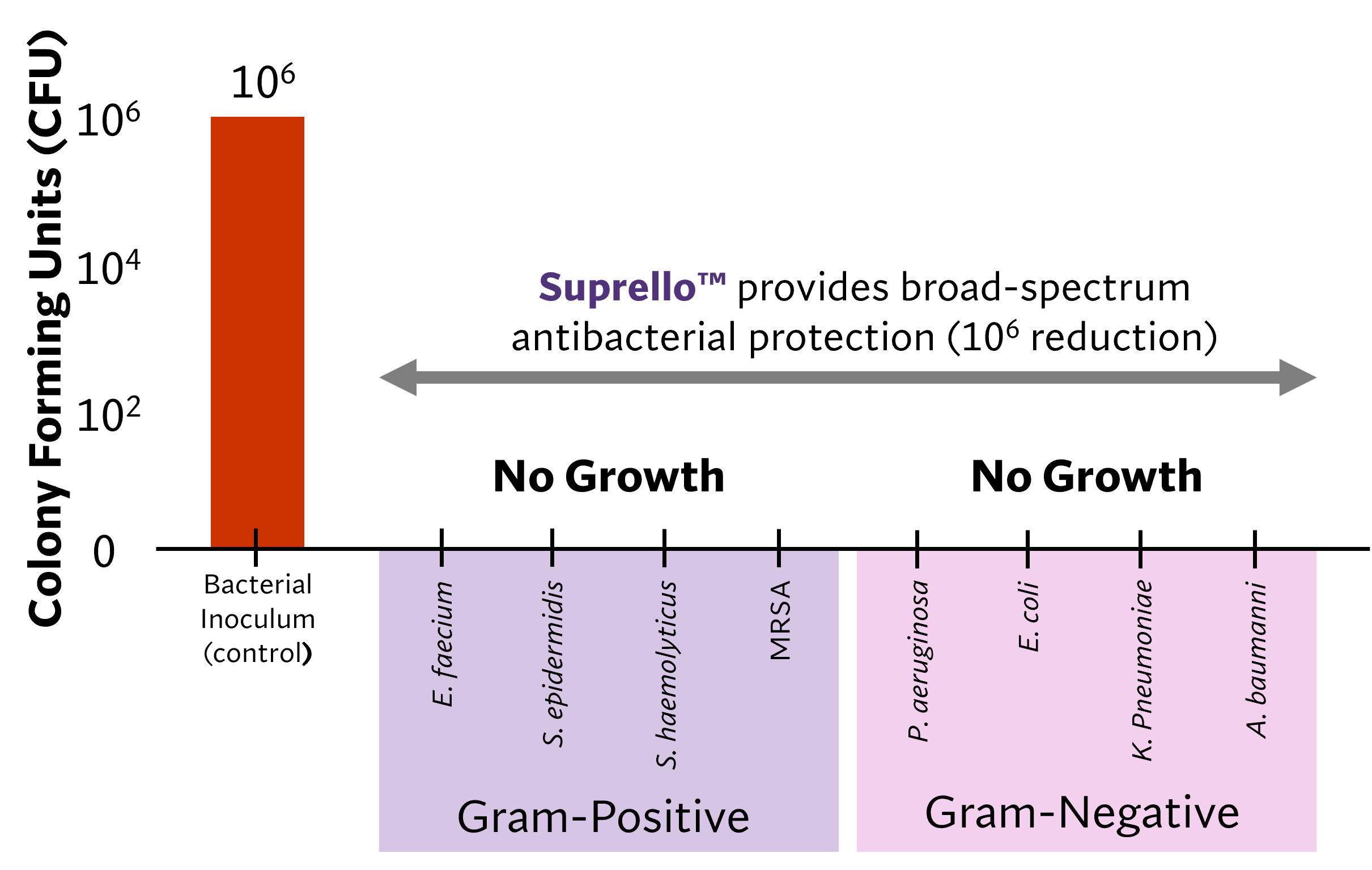
By providing a scaffold for cellular infiltration and capillary growth while also protecting against multidrug-resistant organisms (MDROs)4,5, Suprello™ helps create an optimal environment for wound healing in complex surgical scenarios. Its resorbable matrix conforms to the incision margins to support tissue apposition and wound closure over time6, working in harmony with the body’s natural healing process.
SKU #
G4M100M
GTIN
00850058790060
Brand Name
Suprello™ Surgical Wound Matrix, 2-mL pre-filled syringe with applicator tip
WOUND COVERAGE
20 cm2